Motor Oil 101: Newtonian Fluids versus Non-Newtonian Fluids - Select Synthetics - AMSOIL Authorized Dealer


Main menu:
- Home Page
-
Products
-
AMSOIL Products
- Shop by Product
- Shop by Equipment
- Shop by Category
-
Product Lookup Guides
- Product Lookup Guides - Home
- Filter Lookup Guides
- Auto & Light Truck Lookup Guide
- Motorcycle & Dirt Bike Lookup Guide
- Harley-Davidson Products Guide
- All-Terrain Vehicle Lookup Guide
- Utility Terrain Vehicle Lookup Guide
- Snowmobile Lookup Guide
- Marine Outboard Lookup Guide
- Personal Watercraft Lookup Guide
- Small Engine Lookup Guide
- Free Product Catalog
- AMSOIL - The Blog
- AMSOIL YouTube Videos
- AMSOIL Performance Tests
- The AMSOIL Newsstand
- AMSOIL Testimonials
- AMSOIL Guarantee
- Safety Data Sheets
- Oil Analysis Services
- Shipping & Product Return
- Safe & Secure Shopping
-
AMSOIL Products
- About AMSOIL
-
Oil Basics
- What Is Motor Oil?
- What Is Oil Viscosity?
- What Does Motor Oil Do?
- Why Does Oil Need To Be Changed?
- How Often Should I Change My Oil?
- When Should I Do My 1st Oil Change?
- Can I Mix Different Oils?
- Lubrication Regimes
- Newtonian vs. Non-Newtonian
- Base Oil Groups
- Motor Oil Specifications
- Service Classifications & Grades
- Oil Evaluation Tests
- Oil Filtration
- What is Oil Analysis?
- Why Synthetics?
- Buy Wholesale
- FAQs
- Contact Us
Oil Basics


Newtonian vs. Non-Newtonian
What is the difference between a Newtonian Fluid and a non-Newtonian Fluid?
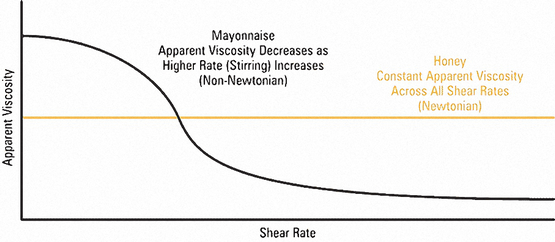
A Newtonian Fluid can be described as a fluid that maintains constant viscosity across all shear rates (shear stress varies linearly with shear rate). These fluids are called Newtonian because they follow the original formula established by Sir Isaac Newton in his “Law of Fluid Mechanics”.
Some fluids, however, don’t behave this way. In general, they are called non-Newtonian Fluids.
The viscosity of non-Newtonian fluids decreases as the shear rate increases (shear thinning), and increases as shear rate decreases (shear thickening).
Imagine you have two jars before you – one filled with mayonnaise, the other filled with honey. Assuming that both jars are affixed to the table’s surface with Velcro, imagine yourself dipping identical butter knives into each of the fluids at the same angle and to the same depth.


Imagine stirring the two fluids by turning the knives at the same speed while keeping the same angle of attack. Which of the two fluids was harder to stir? Your answer should be honey, which is a lot harder to stir than mayonnaise.
Now imagine detaching the jars from the Velcro on the table and turning the jars on their side. Which flows out of the jar faster, honey or mayonnaise? Your answer should be honey; the mayonnaise probably won’t flow at all by simply turning the jar on its side.
Which fluid is more viscous, honey or mayonnaise? If you said mayonnaise, you are correct… at least partially. Likewise, if you said honey you are partially correct.
The reason for this apparent anomaly is that when rotating the knife in both substances, the shear rate varies, while turning each jar on its side is simply measuring the static resistance to flow.
Because honey is a Newtonian fluid while mayonnaise is non-Newtonian, the viscosity of the mayonnaise drops as the shear rate increases, or as the knife is rotated. Stirring subjects the mayonnaise to a high shear stress, causing it to yield to the forcing action.
Conversely, simply setting the jar on its side subjects the mayonnaise to a low shear stress, resulting in little to no viscosity change, so it tends to stay in the jar.
One cannot conventionally measure the viscosity of a non-Newtonian fluid. Rather, one must measure the apparent viscosity, which takes into consideration the shear rate at which the viscosity measurement was made.
Much like viscosity measurements don’t make sense unless the test temperature is reported, apparent viscosity measurements don’t make sense unless the test temperature and the shear rate are reported.
For example, the viscosity of grease is never reported; rather the grease’s apparent viscosity is reported.
(Note: the viscosity may be reported for the base oil used to make the grease, but not of the finished product.)
Generally speaking, a fluid is non-Newtonian if it is comprised of one substance suspended (but not chemically dissolved) in a host fluid. For this to happen, there are two basic categories: emulsions and colloidal suspensions.
An emulsion is the stable physical coexistence of two immiscible fluids. Mayonnaise is a common non-Newtonian fluid, comprised of eggs emulsified into oil, the host fluid. Because mayonnaise is non-Newtonian, its viscosity yields with applied force, making it easy to spread.
A colloidal suspension is comprised of solid particles stably suspended in a host fluid. Many paints are colloidal suspensions. If the paint was Newtonian it would either spread easily but run if the viscosity is low, or spread with great difficulty and leave brush marks, but not run if the viscosity is high.
Because the paint is non-Newtonian, its viscosity yields under the force of the brush, but returns when the brush is taken away. As a result, paint spreads with relative ease, but doesn’t leave brush marks and doesn’t run.
Today's Multigrade Motor Oils – because of their many additives – are also considered non-Newtonian Fluids.


Home Page | Products | About AMSOIL | Oil Basics | Buy Wholesale | FAQs | Contact Us | General Site Map